Our research directions are inspired by natural cellular communication mechanisms and focus on using biotechnology and molecular biology to enable therapeutic development primarily from completely biological materials.
Extracellular Vesicle (Exosome) Biotechnology
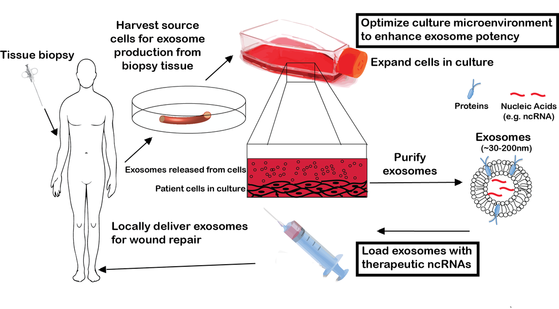
Extracellular vesicles (EVs) - consisting of exosomes, microvesicles, and others - are secreted vesicles of cellular origin that comprise proteins, lipids, and RNAs and are thought to be critical mediators of intercellular communication. These vesicles can function as cell-derived nanoparticles for drug delivery and as alternatives to cell therapies, where their drug-like qualities lend them significant advantages with respect to safety. However, clinical translation of EVs is hindered by limited mechanistic understanding and concerns related to potency, stability, and scalable biomanufacturing. Our group synergizes molecular biology with biomaterials and nanotechnology approaches to engineer EVs for a variety of therapeutic applications.
Recent Publications - EV Gene and Drug Delivery
Recent Publications - EV Gene and Drug Delivery
- Levy D; Abadchi SN; Shababi N; Ravari MM; Pirolli NH; Bergeron C; Obiorah A; Mokhtari-Esbuie F; Gheshlaghi S; Abraham JM; Smith IM; Powsner EH; Solomon TJ; Harmon JW; Jay SM. (2023) Induced pluripotent stem cell-derived extracellular vesicles promote wound repair in a diabetic mouse model via an anti-inflammatory immunomodulatory mechanism. Advanced Healthcare Materials e2300879.
- Levy D; Jeyaram A; Born LJ; Chang K; Abadchi S; Hsu ATW; Solomon T; Aranda A; Harmon JW; Jay SM. (2022) The Impact of Storage Condition and Duration on Bioactivity of Mesenchymal Stromal Cell Extracellular Vesicles. Cytotherapy S1465-3249(22)01025-8.
- Pottash AE; Levy D; Jeyaram A; Kuo L; Kronstadt SM; Chao W; Jay SM. (2022) Combinatorial microRNA loading into extracellular vesicles for increased anti-inflammatory efficacy. ncRNA 8(5):71.
- Kronstadt SM; Jay SM; Jeyaram A. (2022) Extracellular vesicle loading via pH-gradient modification. Methods in Molecular Biology 2504:231-239.
- Kronstadt SK; Pottash AE; Levy D; Wang S; Chao W; Jay SM. (2021) Therapeutic Potential of Extracellular Vesicles for Sepsis Treatment. Advanced Therapeutics
- Born LJ; Chang K; Shoureshi P: Lay F; Bengali S; Hsu ATW; Abadchi S; Harmon JW; Jay SM. (2021) HOTAIR-Loaded Mesenchymal Stem/Stromal Cell Extracellular Vesicles Enhance Angiogenesis and Wound Healing. Advanced Healthcare Materials
- Jeyaram A; Lamichhane TN; Wang S; Dahal E; Kronstadt SM; Levy D; Parajuli B; Knudsen D; Chao W; Jay SM. (2020) Enhanced loading of functional miRNA cargo via pH-gradient modification of extracellular vesicles. Molecular Therapy 28(3):975-985.
- Lamichhane TN; Jay SM. (2018) Production of Extracellular Vesicles Loaded with Therapeutic Cargo. Methods in Molecular Biology 1831:37-47.
- Lamichhane TN; Jeyaram A; Patel DB; Parajuli B; Livingston NK; Arumugasaamy N; Schardt JS; Jay SM. (2016) Oncogene Knockdown via Active Loading of Small RNAs into Extracellular Vesicles by Sonication. Cellular and Molecular Bioengineering Sep;9(3):315-324.
- Lamichhane TN; Raiker RS; Jay SM. (2015) Exogenous DNA Loading into Extracellular Vesicles via Electroporation is Size-Dependent and Enables Limited Gene Delivery. Molecular Pharmaceutics Oct 5;12(10):3650-7.
- Dutta D; Pirolli NH; Levy D; Tsao J; Seecharan N; Xu X; Wang Z; Jia X; Jay SM. (2023) Differentiation state and culture conditions impact neural stem/progenitor cell-derived extracellular vesicle bioactivity. Biomaterials Science
- Kronstadt SM; Patel DB; Born LJ; Levy D; Lerman MJ; Mahadik B; McLoughlin ST; Fasuyi A; Fowlkes L; Hoorens van Heyningen L; Aranda A; Abadchi SN; Chang KH; Hsu ATW; Bengali S; Harmon JW; Fisher JP; Jay SM. (2023) Mesenchymal stem cell culture within perfusion bioreactors incorporating 3D-printed scaffolds enables improved extracellular vesicle yield with preserved bioactivity. Advanced Healthcare Materials e2300584.
- Born LJ; McLoughlin ST; Dutta D; Mahadik B; Jia X, Fisher JP, Jay SM. (2022) Sustained Released of Bioactive Mesenchymal Stromal Cell-Derived Extracellular Vesicles from 3D-Printed Gelatin Methacrylate Hydrogels. Journal of Biomedical Materials Research: Part A
- Patel DB; Luthers CR; Lerman MJ; Fisher JP; Jay SM. (2019) Enhanced extracellular vesicle production and ethanol-mediated vascularization bioactivity via a 3D-printed scaffold-perfusion bioreactor system. Acta Biomaterialia. 95:236-244 Featured in special issue: Cell and Tissue Biofabrication
- Patel DB; Santoro M; Born LJ; Fisher JP; Jay SM. (2018) Towards Rationally Designed Biomanufacturing of Therapeutic Extracellular Vesicles: Impact of the Bioproduction Microenvironment. Biotechnology Advances. 36(8):2051-9.
- Jeyaram A; Jay SM. (2017) Preservation and storage stability of extracellular vesicles for therapeutic applications. AAPS Journal 20(1):1.
- Patel DB; Gray KM; Santharam Y; Lamichanne TN; Stroka KM; Jay SM. (2017) Impact of Cell Culture Parameters on Production and Vascularization Bioactivity of Mesenchymal Stem Cell-Derived Extracellular Vesicles. Bioengineering and Translational Medicine 2(2):170-179.
- Kronstadt SM; Hoorens van Heyningen L; Aranda A; Jay SM. (2023) Assessment of anti-inflammatory bioactivity of extracellular vesicles is susceptible to error via media component contamination. Cytotherapy 25(4):387-396.
- Born LJ; Harmon JW; Jay SM. (2020) Therapeutic Potential of Extracellular Vesicle-Associated Long Non-Coding RNA. Bioengineering and Translational Medicine e10172. Featured in “Futures” special issue.
- Russell AE;...Jay SM;...Vader P. (2019) Biological membranes in EV biogenesis, stability, uptake, and cargo transfer: An ISEV Position Paper arising from the ISEV Membranes and EVs Workshop. Journal of Extracellular Vesicles. 8(1):1684862
- Piard C; Jeyaram A; Liu Y; Caccamese J; Jay SM; Chen Y; Fisher JP. (2019) 3D printed HUVECs/MSCs cocultures impact cellular interactions and angiogenesis depending on cell-cell distance. Biomaterials. 222:119423.
- Thery C; Witwer KW…Jay SM…Zuba-Surma EK. (2019) Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. Journal of Extracellular Vesicles 7(1):1535750.
- Xu J; Feng Y; Conn O; Jeyaram A; Jay SM; Zou L; Chao W. (2018) Circulating plasma extracellular vesicles from septic mice induce inflammation via miRNA- and TLR7-dependent mechanisms. Journal of Immunology 201(11):3392-3400.
- Adams KR; Chauhan S; Patel DB; Clements VK; Wang Y; Jay SM; Edwards NJ; Ostrand-Rosenberg S; Fenselau C. (2018) Ubiquitin conjugation probed by inflammation in MDSC extracellular vesicles. J Proteome Res 17(1):315-324.
- Lamichhane TN; Leung CA; Douti LY; Jay SM. (2017) Ethanol Induces Enhanced Vascularization Bioactivity of Endothelial Cell-Derived Extracellular Vesicles via Regulation of MicroRNAs and Long Non-Coding RNAs. Scientific Reports 7(1):13794.
- Li L; Jay SM; Wang Y; Wu SW; Xiao Z. (2017) IL-12 stimulates CTLs to secrete exosomes capable of activating bystander CD8+ T cells. Scientific Reports 7(1):13365.
- Jay SM; Vunjak-Novakovic G. (2017) Extracellular Vesicles and their Versatile Roles in Tissue Engineering. Tissue Engineering Part A 23(21-22):1210-1211.
- Lamichhane TN; Sokic S; Schardt JS; Raiker RS; Lin JW; Jay SM. (2015) Emerging roles for extracellular vesicles in tissue engineering and regenerative medicine. Tissue Engineering Part B Feb;21(1):45-54.
Bacterial Therapeutics
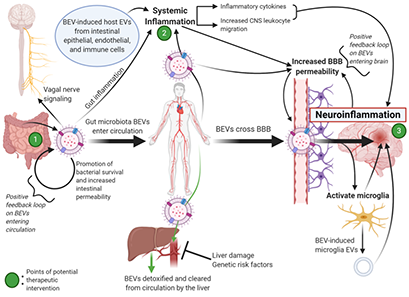
We are exploring the therapeutic potential of live bacteria and bacterial-derived products, especially bacterial extracellular vesicles (BEVs), including the subset of outer membrane vesicles (OMVs). These therapeutics are especially promising for treatment of cancer and neurological diseases and injuries.
Recent Publications
Recent Publications
- Pirolli NH; Reus LSC; Mamczarz Z; Khan S; Bentley WE; Jay SM. (2023) High performance anion exchange chromatography purification of probiotic bacterial extracellular vesicles enhances purity and anti-inflammatory efficacy. Biotechnology and Bioengineering
- Pirolli NH; Bentley WE; Jay SM (2021) Bacterial Extracellular Vesicles and the Gut-Microbiota Brain Axis: Emerging Roles in Communication and Potential as Therapeutics. Advanced Biology 5(7):e2000540
- Hauk P; Stephens K; McKay R; Virgile C; VanArsdale E; Pottash AE; Schardt JS; Jay SM; Sintim HO; Bentley WE. (2020) Homologous quorum sensing regulatory circuit (hQSRC): A dual-input genetic controller for modulating quorum sensing (QS)-mediated protein expression in E. coli. ACS Synthetic Biology 9(10):2692-2702.
- McKay R; Ghodasra M; Schardt JS; Quan D; Pottash AE; Shang W; Jay SM; Payne GF; Chang MW; March JC; Bentley WE. (2018) Genetically Engineered Bacteria as Vehicles for Localized Delivery of Therapeutics: An Application for Crohn’s Disease. Bioengineering and Translational Medicine. 3(3):209-221.
Protein Therapeutics
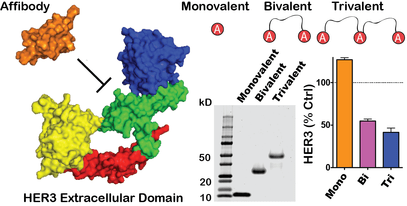
We are interested in designing and developing proteins as therapeutics and drug delivery vehicles. An example is the development of novel multivalent ligands of the ErbB family of receptors, which play prevalent roles in numerous biological processes such as vascularization and cancer. These engineered proteins are capable of inducing specific receptor interactions that lead to therapeutic benefit in various settings, including cardiovascular toxicity and cancer. We continue to utilize protein engineering and design for a variety of purposes, including non-coding RNA delivery, and have interest in applying these approaches to therapeutic applications in wound healing, vascularization, cancer, and more.
Recent Publications
Recent Publications
- Schardt JS; Noonan-Shueh M; Oubaid JM; Pottash AE; Williams SC; Hussain A; Lapidus RS; Lipkowtiz S; Jay SM. (2019) HER3-targeted affibodies with optimized formats reduce ovarian cancer progression in a mouse xenograft model. AAPS Journal. 21(3):48.
- Pottash AE; Kuffner C; Noonan-Shueh M; Jay SM (2019) Protein-based Vehicles for Biomimetic RNAi Delivery. Journal of Biological Engineering. 13:19. Featured in special issue: Emerging Leaders in Biological Engineering
- Schardt JS; Williams SC; Howard JL; Aloimonos CM; Bookstaver ML; Lamichhane TN; Sokic S; Liyasova MS; Hussain A; Lipkowitz S; Jay SM (2017) Engineered Multivalency Enhances Affibody-Based HER3 Inhibition and Downregulation in Cancer Cells. Molecular Pharmaceutics. Apr 3;14(4):1047-1056.
We are grateful to the following agencies/institutions/companies for generously supporting our work:

